Is Naf Covalent Or Ionic
In this commodity, "is NaF ionic or covalent" , the ionic or covalent properties of NaF with detailed explanations are discussed briefly.
Sodium fluoride is an inorganic species with greater ionic character. Information technology shows all the ionic character like any ionic compounds. This salt is water soluble and white colored. Information technology has dissimilar use similar fluoridation of drinking water and pesticides, rat killer and also used to forestall cavities in teeth.
Some questions on sodium fluoride and its chemic behaviour are answered in this article.
Definition of Covalent and Ionic Chemical compound
Covalent and ionic compound are the 2 most important species of Chemistry. All the molecules or chemical species are basically classified into these two compounds.
Sharing of electrons is the keyword of covalent compound considering electronegativity difference between the two atoms is not large. Only in ionic compound complete sharing of electrons have place because of the greater electronegativity divergence betwixt two consisting atoms.

Prototype Credit: Wikipedia
To know more please check: 10 Ionic Bail Examples: Caption And Detailed Facts
How is NaF ionic?
NaF is consisted with two atoms ane is sodium (Na), an electropositive metallic and some other ane is halogen compound fluorine (F), an electronegative element. Electronegativity of sodium is 0.93 (in Pauling calibration) and for fluorine this value is 3.98 (in Pauling scale). In that location is a huge electronegativity difference in sodium and fluorine (=three.05).
The large electronegativity divergence leads NaF to an ionic chemical compound. The atomic number of sodium is 11 (1s2 2sii 2phalf-dozen 3s1) and fluorine is 9 (1s2 2s2 2pfive). Sodium tin can easily donate its 3s electron to fluorine and be unipositive sodium metal ion and gain the nearest noble gas electron configuration. Similarly, fluorine can have the electrons donated by sodium to make the electron configuration like the nearest noble gas.
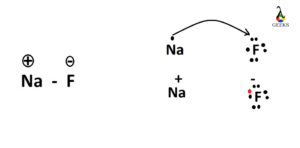
Thus, valence electrons of sodium is completely transferred to fluorine which is the main characteristics of any ionic compound.
% of ionic character (% of I.C) of whatsoever covalent and ionic compound can be determined using the following formula-
% I.C =16 ( χA – χB) + 3.five ( χA – χB)two
In this formula χA and χB are the electronegativity of atom A and B respectively.
Using this formula % of ionic character of sodium fluoride is –
- % I.C = 16 (three.98-0.93) + 3.five (3.98-0.93)2
- % I.C = 81.35
This high value of % of ionic character indicates that NaF is an ionic compound.
To know more please follow: v Polar Covalent Bond Examples: Detailed Insights And Facts
Why NaF is not a covalent compound?
To be a covalent compound, electronegativity difference between 2 molecules must be like or having lesser difference. Thus, electron cloud is shared betwixt two atoms. The electron cloud is shifted more than towards that cantlet having slightly greater electronegativity than the other cantlet.
In NaF, sodium is completely transferring its electrons to fluorine atom every bit fluorine is the well-nigh electronegative element and sodium is 1 of the nearly electropositive elements in periodic table.
From the in a higher place evidence it is clear that NaF is an ionic compound. Too of NaF has a greater amount of lattice energy.
To know more please read: 4 Single Covalent Bond Examples : Detailed Insights And Facts
NaF lewis structure
Lewis structure is invented by Gilbert North. Lewis in 1916 in his article named as "The atom and the molecule". Lewis structure, too known equally electron dot structure, is very much important term to predict the bonded electrons besides equally nonbonding electrons.
To determine the lewis structure of any compound the following steps must be maintained-
- Valance electrons are shown around the corresponding atoms. Nonbonded electrons are non shown.
- Atoms must be continued by unmarried bonds. If the total number of electrons in whatsoever lewis construction and is 'due north' and number of single bonds are 'N', and so (n-2N) number of electrons remain to be placed. These indicate the number of nonbonded electrons.
In this following paradigm the lewis dot structure of water molecule is shown. Oxygen has total eight electrons with 6 valance electrons. Between these half dozen electrons, two electrons form 2 sigma bonds with ii hydrogen atoms and residue of the fours electrons are shown around the oxygen atom in this picture.
Sodium has its valance electron 1 and fluorine has its valence electron v. Sodium donates its 1 valance electron to fluorine and as a consequence fluorine has 6 valance electrons. So, fluorine has at present full 10 and valance electrons vi. These six electrons are shown around the fluorine atom.
To know more please go through: Is Peptide Bond Covalent : How Why, Comparative Analysis and Facts
Frequently Asked Questions (FAQ)
What happens when sodium fluoride reacts with water?
Reply: When sodium fluoride reacts with water, sodium hydroxide (NaOH) and hydrogen fluoride (HF) are formed. NaF + HtwoO → NaOH + HF
Is sodium fluoride soluble in water?
Answer: Sodium fluoride is a polar inorganic compound and h2o is a polar solvent. Thus, sodium fluoride is soluble in water.
Is NaF a basic table salt?
Reply: Yes, sodium fluoride is a bones table salt.
Is Naf Covalent Or Ionic,
Source: https://lambdageeks.com/is-naf-ionic-or-covalent/
Posted by: rosassheaverve02.blogspot.com


0 Response to "Is Naf Covalent Or Ionic"
Post a Comment